PLLR Label Navigation Guide
Label Interpretation Simulator
Learn how to find and read critical safety data on modern drug labels.
Step 1: The Mindset Shift
In the old system, you looked for a letter (A, B, C, D, X). In the PLLR system, you look for Section 8.
Step 2: Navigating Section 8
Section 8 is divided into three critical subsections. Which one do you need?
Pregnancy
Risks to the fetus and clinical data.
Lactation
Breast milk concentrations and effects.
Reproductive Potential
Contraception and pregnancy testing.
Section Analysis
The narrative is broken into three distinct parts. This prevents "generalizing" risk.
The "Bottom Line": What are the actual potential risks?
The Practical Advice: Dosing, timing, and labor impact.
The Evidence: Human studies, animal data, and registries.
The Goal: Benefit-Risk Balancing
You've now moved from a Stop/Go signal (Letter Categories) to a Map (PLLR Narrative).
Key Takeaway:
Instead of avoiding risk entirely, use the narrative to compare the risk of the medication vs. the risk of the untreated illness.
For decades, doctors and patients relied on a simple letter system-A, B, C, D, and X-to figure out if a medication was safe during pregnancy. It seemed straightforward, but it was actually dangerous. The problem? It often created a false sense of security. A drug might be labeled 'Category B' simply because nobody had bothered to study it in pregnant women, not because it was proven safe. This "innocent until proven guilty" bias left healthcare providers guessing and patients at risk.
To fix this, the FDA is the U.S. Food and Drug Administration, the federal agency responsible for protecting public health by ensuring the safety and efficacy of drugs, biological products and medical devices implemented the Pregnancy and Lactation Labeling Rule (or PLLR), a regulatory framework that replaced letter categories with detailed narrative summaries to provide clinically relevant safety data for pregnant and lactating women. Instead of a single letter, you now get a comprehensive story about a drug's risks, the data behind those risks, and how to handle the medication in real-world scenarios. Since millions of pregnancies occur annually in the U.S., and many women take multiple prescriptions, moving from a grade to a narrative is a massive leap in patient safety.
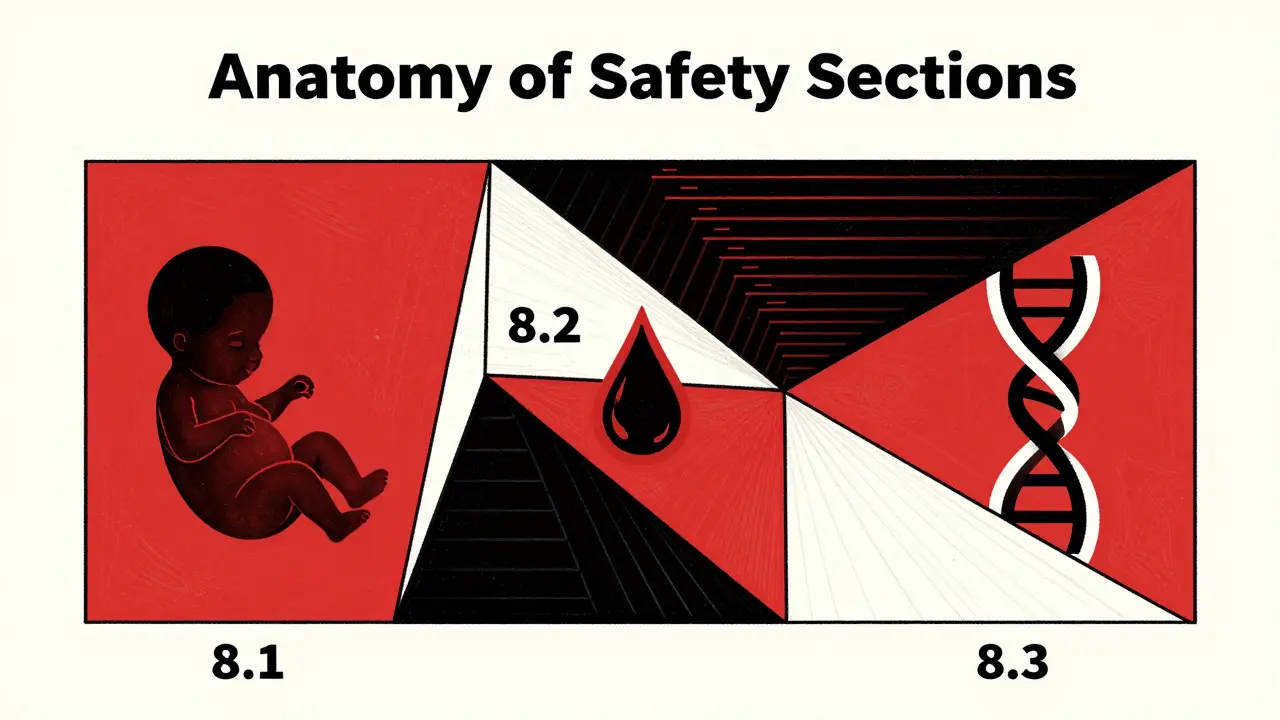
The New Anatomy of Safety Sections
If you open a drug label today, you won't find a letter grade. Instead, look for Section 8: "USE IN SPECIFIC POPULATIONS." The Pregnancy and Lactation Labeling Rule restructured this into three clear subsections: 8.1 (Pregnancy), 8.2 (Lactation), and 8.3 (Females and Males of Reproductive Potential). Each of the first two sections follows a strict three-part format that forces manufacturers to be specific.
- Risk Summary: This is the "bottom line." It describes the potential risks to the fetus, including any known adverse reactions or the level of risk associated with the drug.
- Clinical Considerations: This is where the practical advice lives. It covers dosing alterations, the timing of exposure (which is crucial since different organs develop at different stages), and how the drug affects labor or delivery.
- Data: This provides the evidence. It lists results from human studies, animal data, and the pharmacological activity of the drug.
By separating the "what" (Risk Summary) from the "why" (Data) and the "how" (Clinical Considerations), the FDA ensures that doctors aren't just seeing a warning, but are understanding the context of that warning.
Breaking Down the Pregnancy Subsection
In the old system, if a drug was Category C, it just meant "risk cannot be ruled out." That isn't helpful when a woman is deciding whether to stay on a critical medication for a chronic illness. The new Pregnancy subsection provides a much more nuanced view. For example, it doesn't just say a drug is risky; it might specify that it is associated with oligohydramnios a condition where there is too little amniotic fluid surrounding the fetus, and then explain exactly how the amniotic fluid index changes if the drug is stopped and restarted.
One of the most significant additions is the requirement for Pregnancy Exposure Registries. These are databases that track the outcomes of pregnancies where a specific drug was used. In the past, these were recommended but not mandatory. Now, they are a key part of the labeling process. This shift moves the industry toward active post-marketing surveillance, meaning we get real-world data on how drugs affect development over time, rather than relying solely on small, controlled clinical trials.
Lactation and Reproductive Potential
The PLLR didn't just stop at pregnancy. It overhauled how we view breastfeeding and reproductive health. The Lactation section (8.2) replaces the old "Nursing Mothers" subsection. It now focuses on specific metrics: drug concentrations in breast milk relative to plasma levels and whether the drug interferes with milk production.
Then there is section 8.3, which covers Females and Males of Reproductive Potential. This is a critical area that was previously scattered across labels. It now centralizes three vital pieces of information:
- Pregnancy Testing: When and why a woman needs to be tested to prevent fetal exposure to a toxic drug.
- Contraception: Specific recommendations on birth control, especially if the drug interacts with hormonal contraceptives.
- Infertility: Any known effects the drug has on the ability to conceive.
Comparing the Old Letter System vs. the PLLR
| Feature | Old Letter System (A, B, C, D, X) | PLLR Narrative System |
|---|---|---|
| Format | Single letter grade | Detailed narrative summaries |
| Risk Assessment | Categorical / Generalized | Individualized / Evidence-based |
| Data Transparency | Limited context for the grade | Explicit "Data" subsection provided |
| Update Frequency | Often remained outdated for years | Mandatory updates as new data emerges |
| Focus | Risk avoidance | Benefit-risk balancing |
The Reality of Benefit-Risk Balancing
One of the biggest misunderstandings about drug safety is the idea that the goal is "zero risk." In reality, the goal is the best possible outcome for both the parent and the child. This is where the PLLR truly shines. It allows doctors to consider the risks of an untreated illness.
Imagine a woman with severe depression or a critical heart condition. If a drug is labeled with a known risk, but the risk of her remaining untreated is higher (such as severe maternal stress or organ failure), the narrative format gives the doctor the tools to make a reasoned choice. The letter system was a "stop/go" sign; the PLLR is a map that shows the hazards but also the available routes to safety.

Implementation Pitfalls and Global Differences
Moving to this system wasn't without hiccups. The FDA actually considered using standardized risk statements-similar to how we have "Black Box Warnings"-but they scrapped the idea. Why? Because they realized that standardized phrases would just become the new "letter categories," leading to the same misinterpretations. They decided that a custom, drug-specific narrative was the only way to ensure clarity.
It's also worth noting that this is largely a U.S. approach. When you compare the FDA's method to the European Medicines Agency (or EMA), there is a surprising amount of discordance. Studies have shown that in over 60% of cases, the language used by the EMA and FDA to describe the same drug's pregnancy risk differs significantly. This suggests that interpreting safety sections requires a global perspective, as different regulatory bodies may weigh the same evidence differently.
What happened to the A, B, C, D, X categories?
The FDA officially replaced them with the PLLR in 2014. Because the letter system was often misinterpreted and lacked detail, the FDA required manufacturers to remove these categories and replace them with narrative summaries in the Pregnancy and Lactation sections of the drug labels.
Where do I find the pregnancy safety info on a modern label?
Look for "Section 8: Use in Specific Populations." Within that section, you will find 8.1 for Pregnancy, 8.2 for Lactation, and 8.3 for Reproductive Potential. Each will have a Risk Summary, Clinical Considerations, and a Data section.
Is a "Risk Summary" the same as a warning?
Not exactly. While it does contain warnings, the Risk Summary is designed to provide a balanced view of the risks based on available data. It is meant to be read alongside the "Clinical Considerations" to determine if the benefit of the drug outweighs the risk for a specific patient.
Do all drugs have to follow the PLLR?
The rule applies to prescription drugs and biological products that follow the Physician Labeling Rule (PLR). This includes all products approved since June 30, 2001. For older drugs, the FDA provided a phased timeline to remove the old letter categories.
What is a Pregnancy Exposure Registry?
It is a database that tracks the health outcomes of fetuses and children who were exposed to a specific medication during pregnancy. Under the PLLR, information about these registries is now a mandatory component of the Pregnancy subsection to help fill data gaps.
Next Steps for Healthcare Providers and Patients
If you are a provider, your first step should be to move away from searching for "Category B" or "Category C" in your databases. Instead, focus on the Risk Summary and Clinical Considerations. If the data section is sparse, don't assume the drug is safe-assume the data is missing, and consult a pregnancy exposure registry or a specialized pharmacist.
For patients, the best approach is to ask your doctor: "What is the risk of the medication versus the risk of leaving my condition untreated?" This question forces the conversation into the benefit-risk framework that the PLLR was designed to support. If you are unsure about a drug's safety, you can search for the product's official FDA label to see the full narrative summary for yourself.