Cyclosporine Therapeutic Range & Kidney Function Checker
Analysis Result:
Imagine taking a life-saving medication that protects a new organ but, at the same time, slowly puts the kidneys at risk. That is the tightrope walk of using Cyclosporine is a calcineurin inhibitor immunosuppressant used primarily to prevent organ rejection in transplant recipients. While it is a powerhouse for keeping a transplant healthy, its biggest drawback is cyclosporine nephrotoxicity-the tendency of the drug to damage the very kidneys it is often trying to protect. Because the gap between a dose that works and a dose that causes harm is incredibly small, you can't just "set it and forget it." Precise monitoring is the only way to keep the balance.
Why Cyclosporine Hits the Kidneys
It is a bit of a paradox: the drug prevents the immune system from attacking a graft, but it also changes how blood flows through the kidney. Specifically, it increases vascular resistance in the renal allograft. In plain English, it narrows the blood vessels in the kidney, which can reduce blood flow and lead to acute damage. If levels get too high-typically above 400 ng/ml-structural changes can occur, such as tubular vacuolization and arteriolar hyalinosis.
The good news is that this damage isn't always permanent. Research has shown that nephrotoxicity is often partially reversible if the dose is reduced quickly enough. This is why keeping a close eye on blood levels isn't just a suggestion; it's a requirement for long-term graft survival.
The Gold Standard: Measuring Drug Levels
You can't tell if cyclosporine is at a safe level just by how a patient feels. Doctors rely on Therapeutic Drug Monitoring (TDM), which involves measuring the concentration of the drug in the blood. Not all tests are created equal, and the method used can actually change the result.
For years, laboratories used immunoassays because they are fast and cheap. However, these can sometimes "misread" the blood, picking up metabolites of the drug instead of the active drug itself, leading to falsely high readings. While high-performance liquid chromatography (HPLC) was once the gold standard for accuracy, most modern centers have moved to LC-MS/MS or liquid chromatography-tandem mass spectrometry, which offers nearly 99% specificity. This precision helps doctors avoid dose-adjustment errors that could either lead to organ rejection or kidney failure.
| Method | Accuracy/Specificity | Speed | Main Drawback |
|---|---|---|---|
| Immunoassays | Moderate (10-15% cross-reactivity) | Fast (2-4 hours) | Falsely elevated readings |
| HPLC | High (95-98%) | Slow (6-8 hours) | Expensive equipment |
| LC-MS/MS | Very High (99.2%) | Efficient | Requires specialized lab |
Target Levels: What the Numbers Mean
The "right" level of cyclosporine changes based on the type of transplant and how long the patient has had the organ. Using a one-size-fits-all number is dangerous. For a kidney transplant, the targets usually look like this:
- First week: 200-400 ng/ml (aggressive prevention of early rejection).
- Weeks 2 to 6 months: 125-275 ng/ml.
- 7 to 12 months: 100-150 ng/ml.
- After one year: 75-160 ng/ml.
For heart or liver transplants, the targets are generally higher initially (250-350 ng/ml) before dropping to 100-200 ng/ml after six months. If the levels stay too high for too long, the risk of chronic kidney disease skyrockets.

Tracking Kidney Function in Real-Time
Monitoring the drug level is only half the battle. You also have to monitor how the kidneys are actually performing. If the drug level is "perfect" but the kidney markers are climbing, something is wrong. Doctors typically track a few key markers:
- Serum Creatinine: The primary indicator of kidney function. A target of less than 1.5 mg/dL is generally desired.
- BUN Creatinine Ratio: A ratio under 20:1 helps confirm that the kidney is filtering waste properly.
- Blood Pressure: Since cyclosporine can raise blood pressure, keeping it under 130/80 mmHg is crucial to prevent further kidney strain.
- Magnesium: Levels should ideally stay between 1.7 and 2.2 mg/dL, as the drug can cause magnesium wasting.
C0 vs. C2: The Timing Struggle
When does the blood get drawn? This is a critical detail. Traditionally, doctors measured "trough levels" (C0), which is the lowest concentration of the drug in the blood, measured right before the next dose. While useful, C0 doesn't always tell the whole story.
Many centers now use C2 Monitoring, which is a blood draw exactly two hours after the dose. C2 levels correlate much more strongly with the total drug exposure (the area-under-the-curve). In fact, implementing C2 protocols has been shown to reduce cyclosporine-related kidney toxicity by over 22% because it allows for more precise dosing.
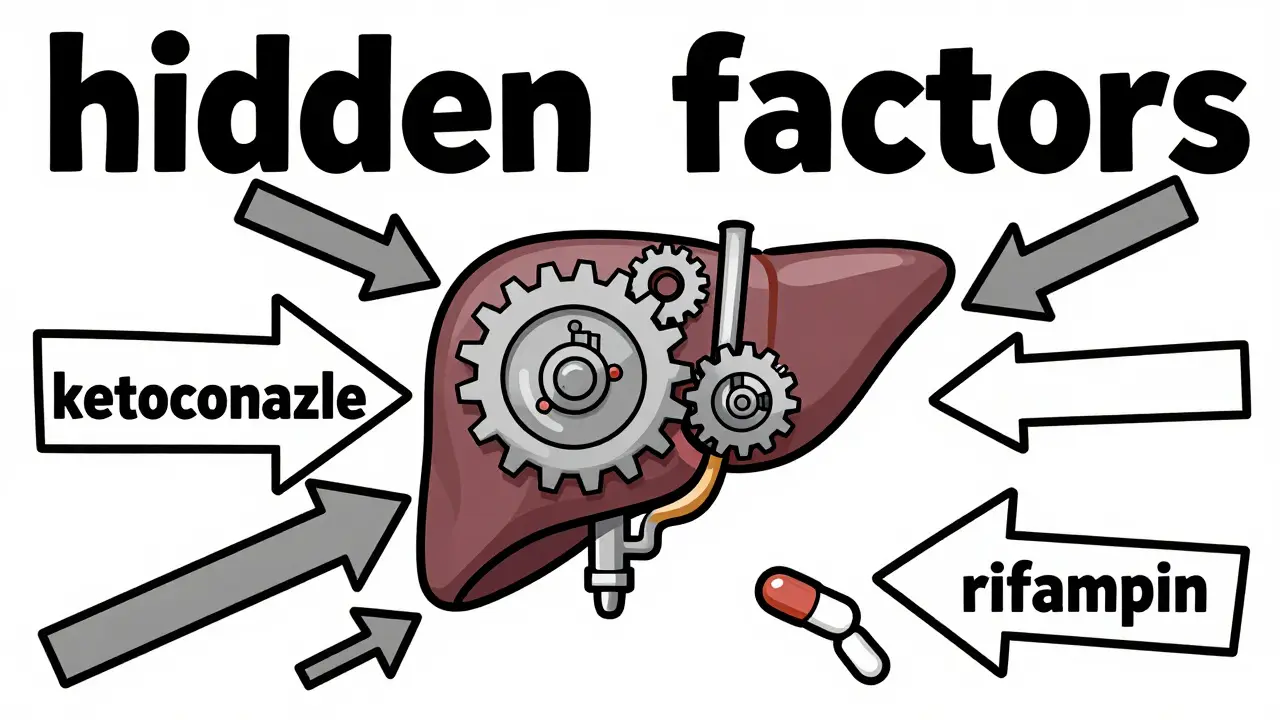
The "Hidden" Factors: Drug Interactions
Cyclosporine doesn't exist in a vacuum. It is processed by the P450 enzyme system in the liver. If a patient starts a new medication that affects these enzymes, their cyclosporine levels can swing wildly without a single dose change.
For example, taking ketoconazole (an antifungal) can spike cyclosporine levels by 30-50%, potentially pushing a patient straight into the nephrotoxic zone. On the flip side, rifampin (an antibiotic) can crash those levels by 40-60%, leaving the transplant vulnerable to rejection. This is why any new prescription must be cross-checked with the immunosuppressant regimen.
Practical Tips for Better Outcomes
Whether you are a clinician or a patient, a few technical details can prevent major errors. First, always use EDTA tubes for blood draws. Using serum separator tubes can cause readings to be 15-20% higher than they actually are, which might lead a doctor to lower a dose unnecessarily.
Second, remember that cyclosporine takes time to stabilize. It can take 4 to 6 weeks to reach a "steady state" after a dose change. Changing the dose every two days based on a single blood test is a recipe for instability. Patience and consistent timing are key.
Can cyclosporine kidney damage be reversed?
Yes, in many cases. Acute nephrotoxicity caused by high doses of cyclosporine is often partially reversible if the dose is reduced or the drug is switched. This is why timely monitoring is so important; the longer the kidneys are exposed to toxic levels, the more likely the damage becomes permanent (chronic).
Why is LC-MS/MS better than a standard immunoassay?
Standard immunoassays can mistake metabolites (breakdown products) of cyclosporine for the active drug, which inflates the results. LC-MS/MS is far more specific, meaning it only measures the active drug, giving the doctor a true reading of the concentration in the blood.
What is the difference between C0 and C2 monitoring?
C0 is the "trough" level, measured just before the next dose. C2 is measured two hours after a dose. C2 is generally considered more accurate for predicting the drug's overall impact on the body and can help reduce the risk of kidney toxicity.
How often should kidney function be checked?
In the first month after a transplant, monitoring is often done twice weekly. This typically drops to once a week between months two and six, and then every two weeks after that. However, this varies based on the patient's stability and the doctor's preference.
Do genetics affect how I respond to cyclosporine?
Yes. Variations in the CYP3A5 gene can change how quickly your body metabolizes the drug. "Extensive metabolizers" break down the drug much faster and may need 30-40% higher doses to keep the drug at a therapeutic level.