Medication Reaction Assessment Tool
This tool helps you evaluate whether your symptoms might be related to medication using the clinical 3-step causality assessment process described in the article.
Did your symptom begin within the typical time frame after taking the medication?
Did stopping the medication improve your symptoms?
Does this symptom match known side effects of the medication?
Assessment Result
When you take a pill, you expect it to help. But sometimes, something unexpected happens - nausea, dizziness, a rash. You might call it a side effect. And you’re not wrong. But here’s the thing: not all bad reactions after taking medicine are side effects. Some are something else entirely - and mixing them up can change how you, your doctor, or even your insurance company handles your care.
What Is a Side Effect?
A side effect is a predictable, known reaction to a drug that happens because of how the drug works in your body. It’s not a mistake. It’s not an accident. It’s just part of the drug’s profile. Think of it like this: if you take a blood pressure medication and feel a little lightheaded when you stand up, that’s a side effect. Why? Because the drug lowers blood pressure - and sometimes, it lowers it too much, too fast. This kind of reaction is common, dose-dependent, and shows up in clinical trials again and again.
The FDA and global health agencies define side effects as unwanted effects that occur at normal doses and are directly linked to the drug’s pharmacology. About 80-85% of all adverse drug reactions fall into this category. These are called Type A reactions. They’re the ones you see in the patient information leaflet: dry mouth from antihistamines, constipation from opioids, or stomach upset from NSAIDs. They’re annoying, sometimes uncomfortable, but rarely dangerous if you’re taking the right dose.
Here’s the key: side effects are confirmed through science. In clinical trials, researchers compare people who took the drug with those who took a sugar pill. If more people in the drug group get a certain symptom - say, headaches - and the difference is statistically significant, then it becomes a documented side effect. For example, in a 2020 JAMA study of the blood thinner apixaban, headache happened in 12.3% of users and 11.8% of placebo users. That tiny difference? Not a side effect. But major bleeding? That happened in 2.1% of apixaban users versus 0.5% in the placebo group. That’s a confirmed side effect.
What Is an Adverse Drug Reaction?
An adverse drug reaction (ADR) is any harmful, unintended response to a medicine taken at normal doses. Sounds like a side effect, right? Not quite. ADRs include side effects - but also things that aren’t predictable at all. These are called Type B reactions. They’re rare, unpredictable, and often have nothing to do with how the drug is supposed to work.
Imagine you take an antibiotic and suddenly break out in hives, your throat swells, and you can’t breathe. That’s not a side effect. That’s an allergic reaction - a Type B ADR. It doesn’t matter how much you took. Even a tiny amount can trigger it. These reactions are not dose-dependent. They’re immune-driven. And they’re not listed in the pamphlet because they’re too rare to show up in trials. They show up later - after thousands of people have used the drug.
Another example: a patient takes statins for cholesterol and develops severe muscle pain. If blood tests show high levels of creatine kinase, and the pain goes away when they stop the drug, then it’s an ADR. But if the pain happens in someone who’s never taken statins before and has no risk factors? That’s probably not related. It’s just a coincidence.

What’s an Adverse Event? (And Why It’s Not the Same)
Now we get to the biggest source of confusion: adverse events. An adverse event is simply any negative health occurrence that happens after you take a drug - whether or not the drug caused it.
Here’s how it works: You take your diabetes medicine. Two days later, you fall and break your hip. You tell your doctor. They log it as an adverse event. But did the medicine cause the fall? Maybe. Maybe not. Maybe you were dizzy from lack of sleep. Maybe your balance was off because of an inner ear infection. The drug might have nothing to do with it. But because it happened after you took the medicine, it gets recorded.
Adverse events are the raw data. They’re the starting point. Side effects and ADRs are what you get after you dig deeper. The Institute for Safe Medication Practices found that 68% of healthcare workers use these terms interchangeably - and that’s dangerous. If every bad thing that happens after taking a pill gets labeled a side effect, doctors start thinking the drug is riskier than it really is. Patients panic. They stop taking life-saving meds. A 2021 study showed 43% of patients quit their prescriptions because they thought every symptom they had was caused by the drug - even when it wasn’t.
Why This Distinction Matters in Real Life
Let’s say you’re on warfarin for a blood clot. You get a headache. You call your pharmacist. They say, “That’s a side effect. Take Tylenol.” But if you’re actually having a brain bleed - a rare, serious ADR - and you think it’s just a headache? That’s how people end up in the ER too late.
Or imagine your insurance denies a claim because your doctor wrote “side effect” when documenting a fall after taking a new antidepressant. The insurer says, “That’s not a side effect. It’s an adverse event - and we don’t cover falls.” Now you’re stuck paying out of pocket.
Even drug labels are changing. The FDA now requires pharmaceutical companies to clearly separate adverse events (all reports) from adverse reactions (confirmed causal links). In 2023, out of 1.2 million adverse event reports submitted to the FDA, only 32.3% were confirmed as actual adverse reactions. That means nearly two-thirds of the reports were coincidences, errors, or unrelated events.
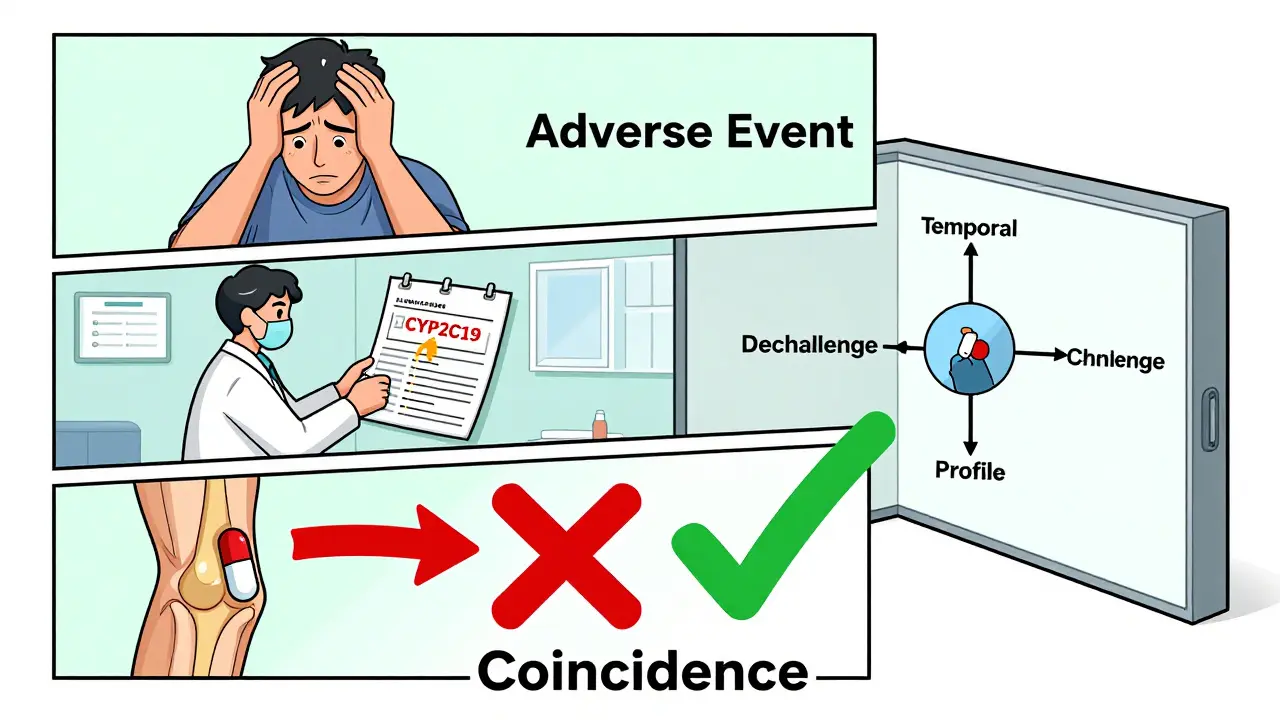
Here’s what top hospitals do: They use a 3-step process to figure out what’s really going on:
- Temporal relationship: Did the symptom start shortly after the drug was taken? (Usually within hours or days.)
- Dechallenge/rechallenge: If you stop the drug and the symptom goes away, then restart it and the symptom comes back - that’s strong evidence.
- Drug profile check: Does this symptom match known side effects in databases like Micromedex? If not, it’s probably not caused by the drug.
Hospitals that use this method saw a 19% drop in medication-related readmissions. That’s real impact.
How Technology Is Helping
It’s not just doctors figuring this out manually anymore. AI tools are now being trained to separate adverse events from true side effects. Companies like ArisGlobal and Oracle have built software that scans patient records, lab results, and clinical notes to flag potential ADRs. In a 2023 study, these tools improved accuracy by 41%.
And it’s getting smarter. Pharmacogenomics - testing your genes to see how you metabolize drugs - is now predicting who’s at risk for certain side effects. For example, people with a specific CYP2C19 gene variant are 8.7 times more likely to have serious stomach bleeding from clopidogrel. That’s not a random side effect. It’s a genetic risk. And now, before prescribing, doctors can test for it.
What You Should Do
You don’t need to become a pharmacologist. But you do need to be smart about how you talk about your symptoms.
- If you feel something new after starting a drug, report it - but don’t assume it’s the drug’s fault.
- Ask your doctor: “Is this a known side effect, or could it be something else?”
- Don’t stop a life-saving medication just because you feel a little off. Talk to your provider first.
- Keep a symptom log: date, time, what you took, what you felt. This helps your doctor spot patterns.
The bottom line: Not every bad feeling after a pill is a side effect. And not every side effect is dangerous. Understanding the difference isn’t just academic - it’s the difference between staying healthy and stopping treatment because you’re scared.
Are all side effects dangerous?
No. Many side effects are mild and temporary - like dry mouth, drowsiness, or upset stomach. These are common and often fade as your body adjusts. But some side effects can be serious, especially if they involve organ damage, severe allergic reactions, or changes in heart rhythm. Always report new or worsening symptoms to your doctor, even if they seem minor.
Can a side effect become an adverse drug reaction?
Not exactly. Side effects are a type of adverse drug reaction - specifically, the predictable, dose-related kind. So it’s the other way around: all side effects are ADRs, but not all ADRs are side effects. Unpredictable reactions like allergies or severe organ damage are ADRs too, but they’re not called side effects because they’re not linked to the drug’s main action.
Why do drug labels list so many side effects if most aren’t dangerous?
Drug labels include every symptom that was statistically more common in clinical trials than in the placebo group - even if it’s mild. This is required by regulators to ensure full transparency. It doesn’t mean you’ll get all of them. Most people experience none, or just one or two. The list is there so you can recognize what’s possible and know when to call your doctor.
If I have an adverse event, does that mean I can’t take the drug again?
Not necessarily. An adverse event is just an occurrence - not proof the drug caused it. If you had a headache after taking a new medicine, but it happened once and never returned, and your doctor rules out other causes, you may still be able to continue. Only confirmed adverse reactions - especially severe ones - usually mean you should stop the drug permanently.
How do I know if my symptom is a side effect or something else?
Look at timing, pattern, and history. Did it start within hours or days of taking the drug? Does it match known side effects? Does it go away when you stop the drug? If yes, it’s likely a side effect. If it’s random, unusual, or happens with other unrelated factors (like stress, infection, or injury), it’s probably not the drug’s fault. When in doubt, talk to your pharmacist or doctor - they can check drug databases and your medical history.







10 Comments
Know the difference or die.
Y'all are out here treating prescriptions like magic beans.
My grandma took warfarin for 12 years and never bled out. She just knew when to call the doc.
Now my cousin thinks a headache = stroke risk. She's off all meds.
It's not the drugs. It's the panic.
And yes, I'm the one who had to explain to her that falling off a ladder isn't a 'drug reaction.'
She still doesn't get it.
But I tried.
Most of those are just people Googling symptoms after taking a pill.
Doc says 'side effect' to cover their ass. Pharmacist says 'side effect' so you don't sue.
Insurance says 'not a side effect' so they don't pay.
Everyone's lying.
But the truth? It's messy.
And nobody wants to admit that.
This is not opinion. This is clinical pharmacology 101.
Hospitals that implement this protocol reduce readmissions by 19%.
That's not anecdotal. That's peer-reviewed.
Why are we still treating symptoms like causality?
It's a systemic failure in medical literacy.
my doc said it wasnt the med.
but i was so scared i didnt trust him.
now i take it again.
but i keep a log.
and i cry every time i swallow the pill.
im not okay.
What?
Since when?
I thought every bad feeling after a pill was the pill's fault.
Are you telling me my insomnia from Zoloft isn't a side effect?
It's an adverse event?
Then what's a side effect?
Do I need a PhD?
My mom stopped her diabetes med because she thought her toe numbness was 'the drug' - turned out it was a pinched nerve from her new shoes.
She's back on meds now.
And we laugh about it.
But imagine if she didn't talk to her pharmacist? 😅
Then I got sick.
Real sick.
And I needed the very drugs I was terrified of.
Learning the difference between side effect and ADR saved my life.
Not because I became a doctor.
But because I stopped panicking.
And started asking better questions.
You don't need to be an expert.
You just need to be curious.
And not assume the worst.
Patients come in saying 'I got a rash after the pill' - and we find out they were hiking in poison ivy the day before.
Or they say 'the antidepressant made me dizzy' - turns out they didn't sleep in 3 days.
It's not the drug. It's life.
But we still have to document it.
And we still have to listen.
Just... gently.
And with a database open. 🙏
Patients report 'side effects' for everything.
Then when we check the data - 80% are unrelated.
But we still have to log them.
Because one day, that one outlier?
It's the signal.
And someone's life depends on it.
So we listen.
Even when it's noise. 🇬🇧